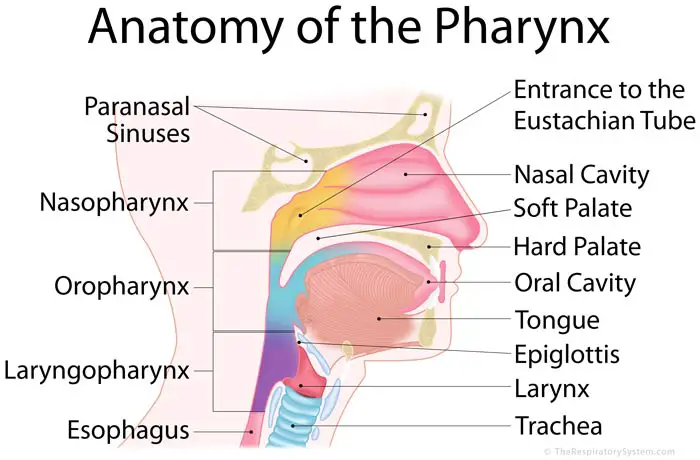
The eardrum separates this space from the ear canal. Is tympanic cavity and middle ear cavity same?Īlso known as the tympanic cavity, the middle ear is an air-filled, membrane-lined space located between the ear canal and the Eustachian tube, cochlea, and auditory nerve. Also known as the auditory tube, it helps keep the middle ear healthy by equalizing pressure, clearing secretions, and protecting it from pathogens that might otherwise cause infections. The eustachian tube extends from the middle ear to the upper part of the throat behind the nose. The Eustachian tube measures only 17-18mm and is horizontal at birth. The Eustachian tube opens into the nasopharynx. The function of this tube is to protect, aerate and drain the middle ear (and mastoid). This generic type of device includes the esophageal dilator tracheal bistour (a long, narrow surgical knife) tracheal dilator tracheal hook laryngeal injection set laryngeal knife laryngeal saw laryngeal trocar laryngectomy tube adenoid curette adenotome metal tongue depressor mouth gag oral screw salpingeal curette tonsillectome tonsil guillotine tonsil screw tonsil snare tonsil suction tube tonsil suturing hook antom reforator ethmoid curette frontal sinus-rasp nasal curette nasal rasp nasal rongeur nasal saw nasal scissors nasal snare sinus irrigator sinus trephine ear curette ear excavator ear rasp ear scissor, ear snare ear spoon ear suction tube malleous ripper mastoid gauge microsurgical ear chisel myringotomy tube inserter ossici holding clamp sacculotomy tack inserter vein press wire ear loop microrule mirror mobilizer ear, nose, and throat punch ear, nose and throat knife and ear, nose, and throat trocar.Auditory tube: The tube that runs from the middle ear to the pharynx, also known as the Eustachian tube. An ear, nose, and throat manual surgical instrument is one of a variety of devices intended for use in surgical procedures to examine or treat the bronchus, esophagus, trachea, larynx, pharynx, nasal and paranasal sinus, or ear. If FDA promulgates a regulation under section 515(b) of the act requiring premarket approval for a device, section 501(f)(1)(A) of the act applies to the device. Accordingly, unless an effective date of the requirement for premarket approval is shown in the regulation for a device classified into class III in this part, the device may be commercially distributed without FDA's issuance of an order approving a PMA declaring completed a PDP for the device. Such a regulation under section 515(b) of the act shall not be effective during the grace period ending on the 90th day after its promulgation or on the last day of the 30th full calendar month after the regulation that classifies the device into class III is effective, whichever is later. (a ) Before FDA requires that a device commercially distributed before the enactment date of the amendments, or a device that has been found substantially equivalent to such a device, has an approval under section 515 of the act FDA must promulgate a regulation under section 515(b) of the act requiring such approval, except as provided in paragraph (b) of this section. Nasopharyngoscope (flexible or rigid) and accessories.Įar, nose, and throat drug administration device.Įar, nose, and throat examination and treatment unit.Įxternal upper esophageal sphincter compression device. Powered insertion system for a cochlear implant electrode array.Īrgon laser for otology, rhinology, and laryngology.Įar, nose, and throat microsurgical carbon dioxide laser.īronchoscope (flexible or rigid) and accessories.Įsophagoscope (flexible or rigid) and accessories. Transcutaneous air conduction hearing aid system.Įar, nose, and throat electric or pneumatic surgical drill.Įar, nose, and throat fiberoptic light source and carrier.Įar, nose, and throat manual surgical instrument. Tympanostomy tube with semipermeable membrane. Prosthesis modification instrument for ossicular replacement surgery.Įar, nose, and throat synthetic polymer material. Partial ossicular replacement prosthesis. Group hearing aid or group auditory trainer.Īctive implantable bone conduction hearing system. 
Hearing aid calibrator and analysis system. Short increment sensitivity index (SISI) adapter.Įarphone cushion for audiometric testing.Įlectronic noise generator for audiometric testing. Limitations of exemptions from section 510(k) of the Federal Food, Drug, and Cosmetic Act (the act).Īcoustic chamber for audiometric testing. Effective dates of requirement for premarket approval.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
